Initial Results and Trials in Process - NCI MyeloMATCH Presentations at ASH 2025
- Posted:
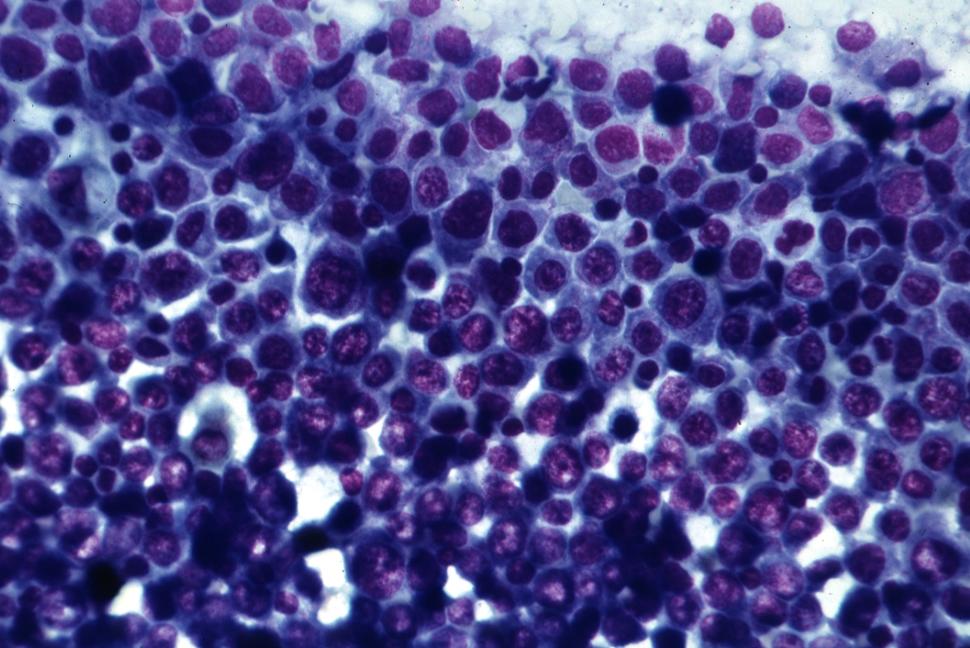
Acute myeloid leukemia cells with characteristics indicating recent transition from myeloproliferative disorder.
Credit: University of Alabama at Birmingham Department of Pathology
MyeloMATCH (Myeloid Malignancies Molecular Analysis for Therapy Choice) is a group of precision medicine clinical trials for people with myelodysplastic syndromes (MDS) or acute myeloid leukemia (AML).
The MyeloMATCH screening trial and five treatment trials of targeted combination agents are open for patient enrollment, with more treatment trials planned. The unique design of MyeloMATCH allows patients to be followed along the course of their disease, with possible additional treatment options available to them depending on further genetic testing.
MyeloMATCH opened in mid-2024, and the first results from the trial are being presented at the 2025 ASH Annual Meeting (December 6-9, 2025; Orlando, FL). The following oral presentation and five posters will be presented.
Oral Abstract Session
Identifying fusions in AML/MDS: An experience from first 400 patients enrolled into MyeloMATCH
December 6; 4:45-5:00 pm EST
ABSTRACT ID 340
Poster Abstract Sessions
A randomized Phase II trial of ASTX727 and venetoclax with or without enasidenib for newly diagnosed older adults with IDH2 mutant Acute Myeloid Leukemia: A MyeloMATCH substudy (MM1OA-S03)
December 7; 6:00 – 8:00 pm EST
ABSTRACT ID 3466
Improving karyotype success for MyeloMATCH: Evidence-based culturing optimization for rapid AML screening - an NCI MyeloMATCH and SWOG report
December 7; 6:00 – 8:00 pm EST
ABSTRACT ID 3473
Chromosome genomic array testing (CGAT) improves diagnostic yield in patients with AML and MDS and has the potential for better risk stratification - an NCI MyeloMATCH and SWOG report
December 7; 6:00 – 8:00 pm EST
ABSTRACT ID 3501
Biallelic TP53 aberrations and double TP53 mutations are prevalent in AML/MDS patients with del(5q) complex karyotype - an NCI MyeloMATCH and SWOG report
December 8; 6:00 – 8:00 pm EST
ABSTRACT ID 5276
Myeloid malignancies molecular analysis for therapy choice (MyeloMATCH): Rapid availability of clinical, pathologic and molecular features for treatment assignment in the first 550 patients.
December 8; 6:00 – 8:00 pm EST
ABSTRACT ID 5279
“The goal of MyeloMATCH is to test combinations of drugs to treat the disease in a highly targeted way and to be able to start treatment quickly after diagnosis,” said NCI’s coordinator for the trial, Richard F. Little, MD. “We’re pleased to share these early results on the first several hundred patients who were screened.”
MyeloMATCH is being conducted by the NCI National Clinical Trials Network, with participation of the NCI Community Oncology Research Program, the Frederick National Laboratory for Cancer Research, Fred Hutch Cancer Center, and Children’s Hospital Los Angeles. The SWOG Cancer Research Network is coordinating the screening protocol.